Luo, Z., et al. Developing a Next-Generation Vaccine Adjuvant System through Rational Design to Alleviate AS01 Adjuvant Deficiency. J Med Chem, 2026 Mar 19, PMID: 41855509
- This research addresses the critical need to improve vaccine adjuvants, which are
essential for enhancing immune responses in subunit vaccines. While AS01 has
demonstrated potent immunostimulatory properties, its widespread application is
hindered by the scarcity and toxicity of QS-21, a key component. Additionally, the
liposomal formulation of AS01 imposes storage challenges requiring strict
refrigeration, limiting its practicality. Developing safer, more cost-effective, and
storage-friendly adjuvants is therefore vital for expanding vaccine accessibility and
effectiveness, especially in resource-limited settings. This study's focus on creating
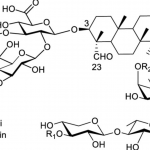
novel nonliposomal adjuvants, BST01 and BST02, leveraging simplified QS-21
derivatives, aims to overcome these limitations, potentially facilitating broader
vaccine deployment with comparable efficacy and improved safety profiles. - The main results demonstrate that the novel nonliposomal adjuvants BST01 and
BST02, formulated by combining simplified QS-21 derivatives (VA05 and VA06) with
MPLA, exhibit comparable immunogenicity to the traditional AS01b adjuvant but with
significantly improved safety profiles, notably reduced hemolytic toxicity. Cryo-TEM
imaging revealed that both BST01 and BST02 can self-assemble into nanowire
structures, facilitating effective delivery and lymphatic drainage of immune
components. In vivo studies confirmed that these adjuvants induce robust antigen-
specific antibody responses and cellular immunity comparable to QS-21-based
systems, with the added advantage of enhanced stability and simplified
manufacturing without reliance on liposomal formulations. Collectively, these findings
may position BST01 and BST02 as promising next-generation vaccine adjuvants
capable of addressing the limitations of current formulations. - Comment from DK: The study's limitations suggest that further research, including
clinical trials, is needed to validate the efficacy, safety, and practical application of
BST01 and BST02 in humans. These limitations include the following: Preclinical
Focus: The research primarily involves in vitro experiments and mouse models, which may not fully replicate human immune responses, necessitating further clinical
validation. Limited Scope of Antigens: The immunogenicity assessments are
conducted with specific model antigens (OVA, VZV-gE, Mtb-M72), and it remains to
be seen whether the adjuvants are equally effective across a broader range of
pathogens and vaccine formulations. Short-Term Evaluation: The immune responses
are monitored over relatively short periods post-immunization, leaving the long-term
durability of the induced immunity unaddressed. Potential Translational Challenges:
While the adjuvants show promising stability and safety profiles in animal models,
scalability, manufacturing consistency, and regulatory hurdles for human application
are still to be considered. Mechanistic Insights: Although the structural and
transcriptomic analyses provide insights into the adjuvants’ mechanisms, detailed
molecular pathways and cellular interactions remain to be elucidated
comprehensively.
Click here to access the full scientific paper.
Recent Posts
 Kinsey, C., et al. A Versatile Reversed-Phase Liquid Chromatography Charged Aerosol Detection Method for Streamlined Monitoring of QS-21 Content and Stability in Liposomal Adjuvant Formulations. https:// doi.org/10.26434/chemrxiv.15002275/v1. Posted on 21 April 2026 (this preprint has not been peer reviewed. Data may be preliminary)
Kinsey, C., et al. A Versatile Reversed-Phase Liquid Chromatography Charged Aerosol Detection Method for Streamlined Monitoring of QS-21 Content and Stability in Liposomal Adjuvant Formulations. https:// doi.org/10.26434/chemrxiv.15002275/v1. Posted on 21 April 2026 (this preprint has not been peer reviewed. Data may be preliminary)