San Martín, R., et al. Structure and immunological activity of QS-21 variant from Quillaja saponaria aerial biomass. Front Immunol, 2026 Mar 9, 17:1771912, PMID: 41878432
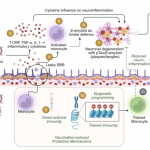
- This study focuses on QS-21, a potent saponin adjuvant essential for human
vaccines, traditionally sourced from the bark of mature Quillaja saponaria trees in
Chile. The researchers identified a natural variant, QS-21-Rha, predominantly found
in the leaves and twigs of young California-grown shrubs, which can be extracted
with high purity. Importantly, QS-21-Rha exhibits immunological activity comparable
or superior to bark-derived QS-21, particularly in activating CD4+ T cells. This
discovery offers a promising, eco-friendly, and scalable source of vaccine adjuvant,
potentially enhancing supply chain resilience, reducing environmental harm, and
lowering manufacturing costs, which are crucial factors for expanding global vaccine
access. - The main results of the study demonstrate that QS-21-Rha, a naturally occurring
variant isolated from the leaves and twigs of young Quillaja saponaria shrubs grown
in California, can be produced in high purity and yields from renewable plant
material. Structurally, QS-21-Rha differs from traditional bark-derived QS-21 by the
substitution of rhamnose at the C3 position of the glycoside moiety. Immunologically,
QS-21-Rha formulated into liposomes elicits robust adaptive immune responses,
notably strong activation of CD4+ T cells and increased cytokine production, often
exceeding the responses induced by traditional QS-21. Additionally, the production
of QS-21-Rha requires significantly less biomass and is more environmentally
sustainable, offering advantages in scalability, quality control, and cost-effectiveness
for vaccine manufacturing. - Comment from DK: Despite the advantages of QS-21-Rha described in this paper,
the traditional QS-21 has been extensively characterized and has an established
safety and efficacy profile based on numerous preclinical and clinical studies. Its
well-understood mechanisms of action, stability, and compatible formulation with
existing vaccine platforms provide a significant advantage in translational
development and regulatory approval processes. Additionally, the complex mixture
of isomers present in traditional bark-derived QS-21 may offer a broader range of
immunostimulatory properties, which could be beneficial for certain vaccine
formulations or disease targets. The single-isomer composition and defined
chemical structure of QS-21-Rha could potentially limit some of the broader
immunostimulatory effects that the mixture of isomers in traditional QS-21 might
confer. Moreover, as the field is more familiar with the long-term safety and
immunogenicity data of traditional QS-21, regulatory pathways may be more
straightforward for its use, whereas QS-21-Rha would require additional validation.
In summary, the traditional QS-21's comprehensive safety profile, established
efficacy, and formulation stability may make it still preferable in certain contexts,
even if QS-21-Rha shows promising immunological benefits.
Click here to access the full scientific paper.
Recent Posts
 Kinsey, C., et al. A Versatile Reversed-Phase Liquid Chromatography Charged Aerosol Detection Method for Streamlined Monitoring of QS-21 Content and Stability in Liposomal Adjuvant Formulations. https:// doi.org/10.26434/chemrxiv.15002275/v1. Posted on 21 April 2026 (this preprint has not been peer reviewed. Data may be preliminary)
Kinsey, C., et al. A Versatile Reversed-Phase Liquid Chromatography Charged Aerosol Detection Method for Streamlined Monitoring of QS-21 Content and Stability in Liposomal Adjuvant Formulations. https:// doi.org/10.26434/chemrxiv.15002275/v1. Posted on 21 April 2026 (this preprint has not been peer reviewed. Data may be preliminary)